
By Randi Kudner, ASTRO Senior Quality Improvement Manager
Radiation oncologists and other physicians are struggling to maximize return on investment under the new Merit-based Incentive Program Payment System (MIPS), compounding questions about the new program’s burden and value.
The Medicare Access and CHIP Reauthorization Act of 2015 (MACRA) established the Quality Payment Program (QPP) to transition Medicare payment away from fee-for-service to pay-for-performance. Participation in the QPP is through one of two pathways. The Advanced Alternative Payment Model (APM) is one option; however, only radiation oncologists in a multidisciplinary practice involved in the Oncology Care Model currently have this option. A radiation oncology APM is currently in development.
The Merit-based Incentive Payment System (MIPS) is the other option, and it is how most radiation oncologists currently report. MIPS combines and replaces Physician Quality Reporting System (PQRS), Value-based Payment Modifier (VM) and Medicare EHR Incentive (Meaningful Use) programs into one comprehensive program.
The Center for Medicare and Medicaid Services (CMS) recently released feedback data for the 2017 MIPS performance year. Included in the feedback report is the:
As we work to influence the 2019 QPP proposed rule, we are also interested in gathering performance data from our members so that we may advocate effectively on theirn behalf. What was the participation rate? What were the scores? How did our members perform?
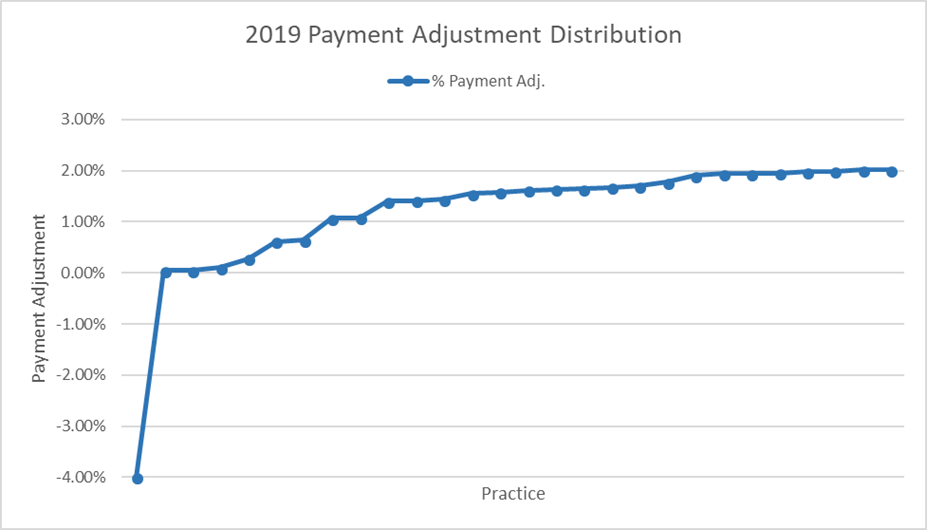
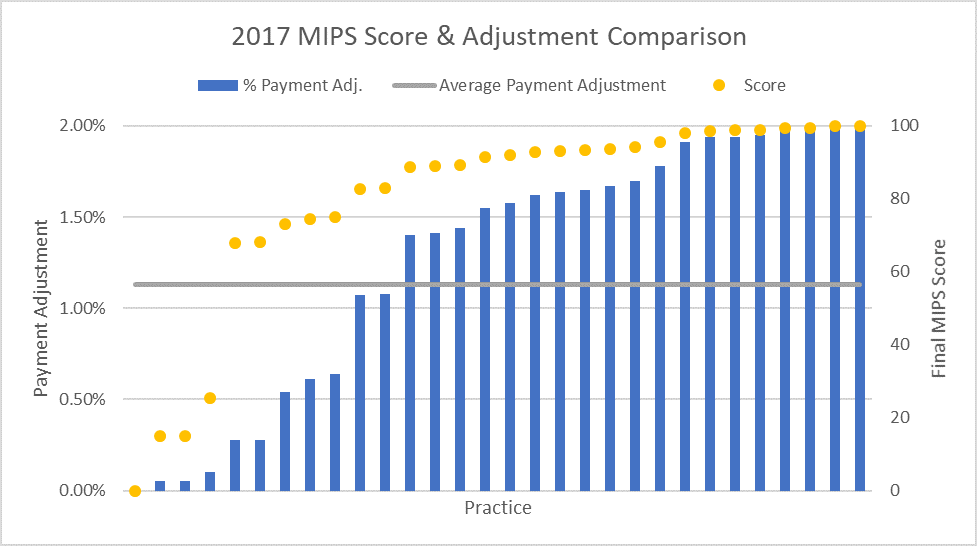
ASTRO requested feedback data from some groups and has some initial data, but we need more responses to advocate for program improvements. The 2017 program included up to a positive or negative 4 percent payment adjustment for 2019, and many ASTRO members that were initially polled scored a perfect 100. However, when all calculations were averaged, this only equates to a 2.02 percent positive payment adjustment in 2019.
Radiation oncologists and other physicians are struggling to maximize return on investment under the new Merit-based Incentive Program Payment System (MIPS), compounding questions about the new program’s burden and value.
The Medicare Access and CHIP Reauthorization Act of 2015 (MACRA) established the Quality Payment Program (QPP) to transition Medicare payment away from fee-for-service to pay-for-performance. Participation in the QPP is through one of two pathways. The Advanced Alternative Payment Model (APM) is one option; however, only radiation oncologists in a multidisciplinary practice involved in the Oncology Care Model currently have this option. A radiation oncology APM is currently in development.
The Merit-based Incentive Payment System (MIPS) is the other option, and it is how most radiation oncologists currently report. MIPS combines and replaces Physician Quality Reporting System (PQRS), Value-based Payment Modifier (VM) and Medicare EHR Incentive (Meaningful Use) programs into one comprehensive program.
The Center for Medicare and Medicaid Services (CMS) recently released feedback data for the 2017 MIPS performance year. Included in the feedback report is the:
- 2017 final score
- 2019 MIPS payment adjustment
- Final performance category scores and weights
As we work to influence the 2019 QPP proposed rule, we are also interested in gathering performance data from our members so that we may advocate effectively on theirn behalf. What was the participation rate? What were the scores? How did our members perform?
ASTRO requested feedback data from some groups and has some initial data, but we need more responses to advocate for program improvements. The 2017 program included up to a positive or negative 4 percent payment adjustment for 2019, and many ASTRO members that were initially polled scored a perfect 100. However, when all calculations were averaged, this only equates to a 2.02 percent positive payment adjustment in 2019.
There was little surprise that the positive payment adjustments were lower than the 4 percent ceiling, considering the budget neutral mandate combined with the "Pick Your Pace" option introduced to ease practices into the new program. Yet the fact that the positive adjustment was so low—about half of the maximum—means that some program rules should be reviewed. Realistically, if the program is to succeed, there should be a meaningful financial benefit, alongside the potential quality improvement impact, that offsets the significant cost of participation.
Clinicians and groups can access their 2017 MIPS performance feedback on the QPP website using an Enterprise Identity Management (EIDM) account. The CMS Guide to Obtaining an EIDM Account provides instructions on how to create one. If an error was made in the calculation of your 2019 MIPS payment adjustment, you should request a targeted review from CMS via your EIDM account on the QPP website. Targeted reviews can be submitted until October 1, 2018. Circumstances warranting such a request include:
- Errors or data quality issues on submitted measures and activities;
- Physician eligibility questions that could, for instance, be related to CMS’s low-volume threshold and that would preclude a payment adjustment;
- An erroneous exclusion from the alternative payment model participation list; or
- Failure to be automatically reweighted due to eligibility provided by CMS’s 2017 extreme and uncontrollable circumstances policy.




 RSS Feed
RSS Feed